Unique Device Identification (UDI) for Medical Device
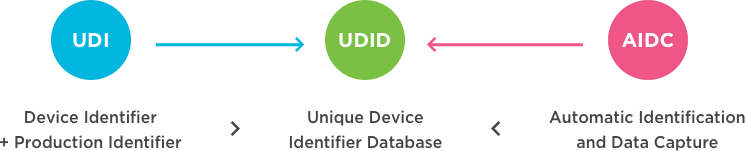
What is UDI?
A Unique Device Identification (UDI) system is intended to provide single, globally harmonized positive identification of medical devices through distribution and use, requiring the label of devices to bear a globally unique device identifier (to be conveyed by using Automatic Identification and Data Capture and, if applicable, its Human Readable Interpretation) based upon standard, with the UDI-DI (Device Identifier) of that unique identifier being also linked to a jurisdiction-specific public UDI database (source: International Medical Device Regulators Forum (IMDRF): http://www.imdrf.org/).
How GS1 Standards can be used for UDI?
GS1 is accredited as an UDI Issuing Agency/Entity based on many regulations worldwide, in particular US, EU, China, South Korea, Saudi Arabia, meaning that manufacturers supplying regulated medical devices to these markets can use the GS1 standards to implement the UDI requirements.
The GS1 system of standards provides a global framework to identify, capture and share Healthcare product information, thereby enabling a consistent worldwide implementation of UDI. UDI regulatory requirements have a translation into GS1 standards as shown in the table.
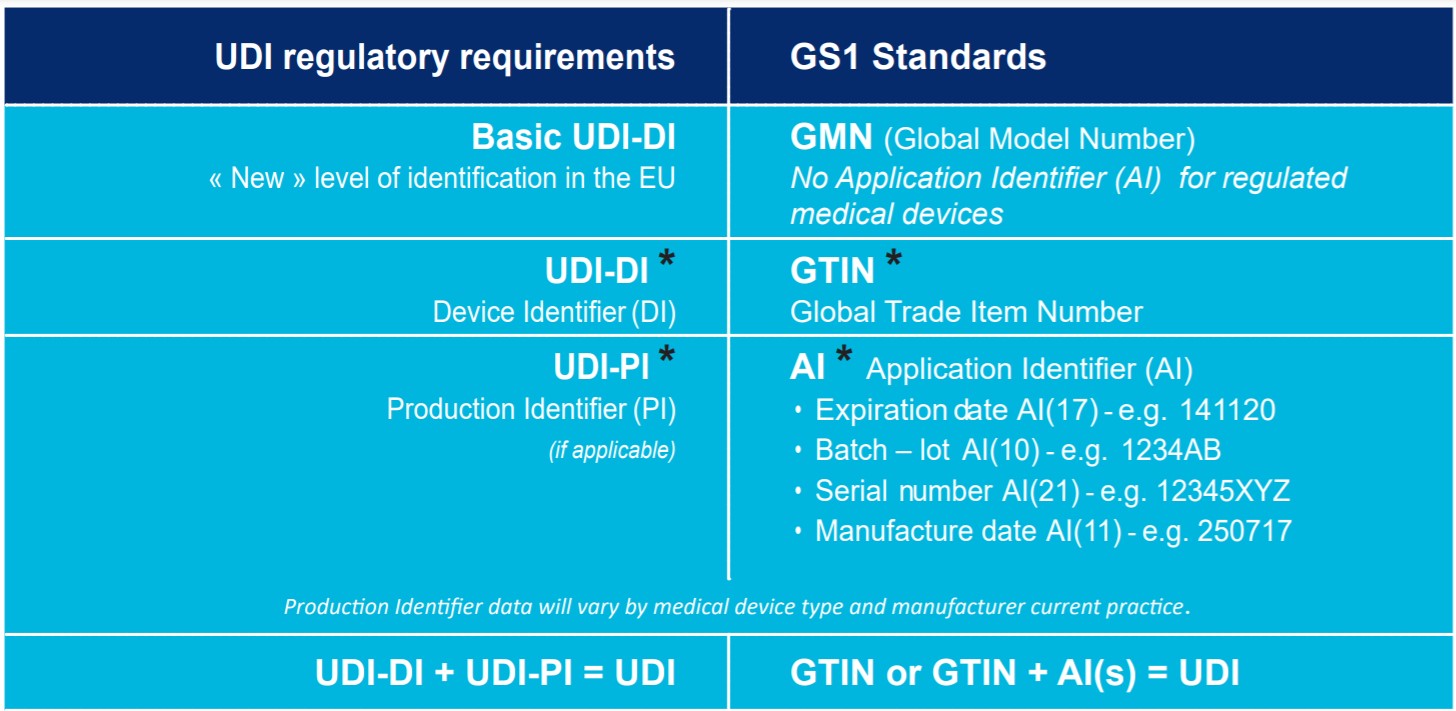
* The HRI Format shall follow the rules of the UDI Issuing Entity
GS1 Standards for UDI in the EU
In difference to the US FDA regulation, the EU regulations introduce a new identifier – the “Basic UDI-DI”. It allows to group medical devices with similar features within the EU regulatory database EUDAMED. It is assigned outside of the normal trade item supply chain. The assignment must be done by the medical devices manufacturer or authorised representative, before the product can be submitted for market registration/approval to the competent authorities.
Composition of the GMN (Basic UDI-DI)
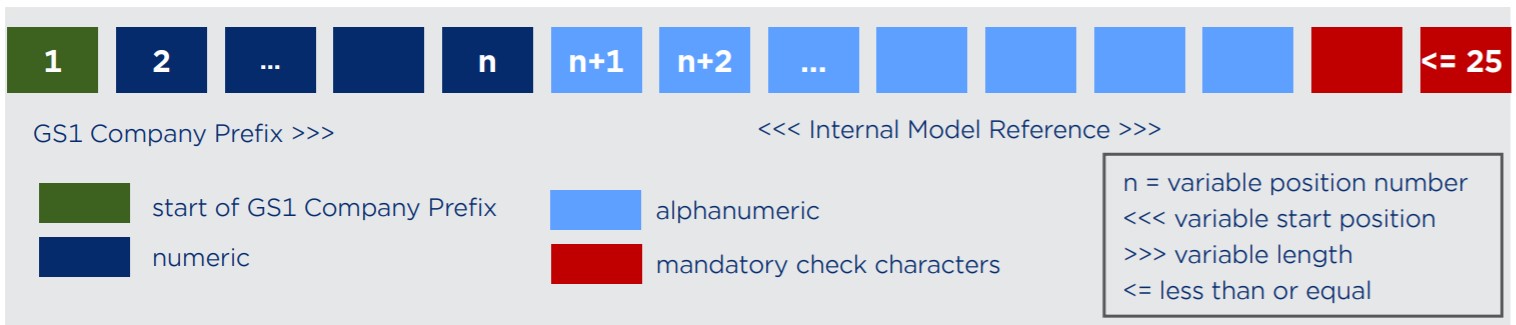
To calculate the GMN check characters:
https://www.gs1.org/services/gmn-generator
Composition of the GTIN (UDI-DI)
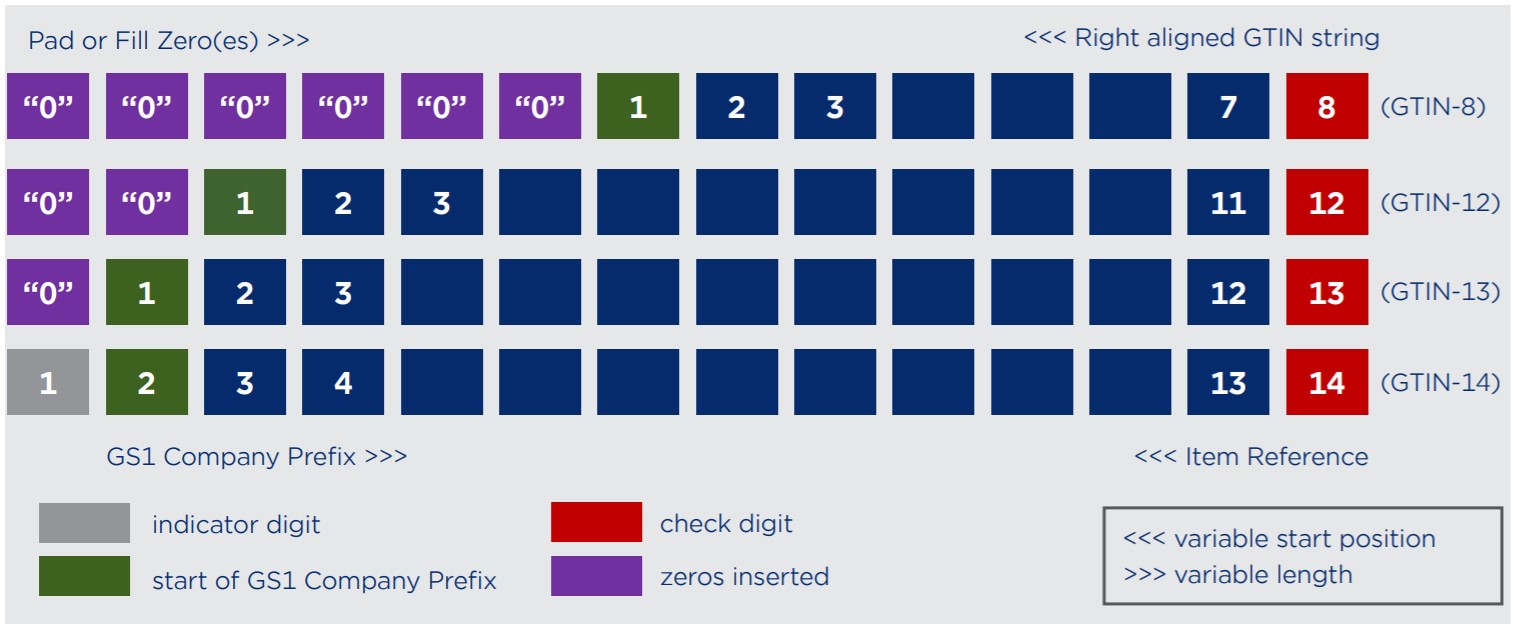
To calculate the GTIN check digit: https://www.gs1.org.sg/Resources/Tools/CheckDigitCalculator.aspx